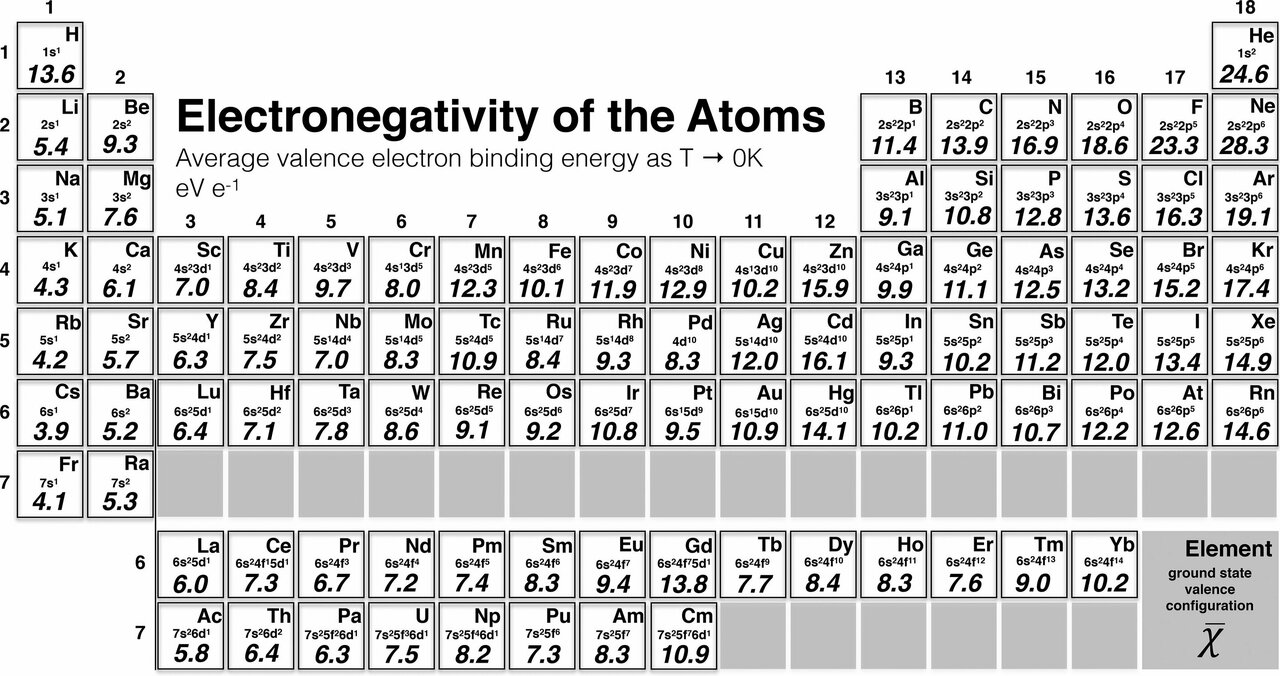

Electronegativity has no units but "Pauling units" are often used when indicating values mapped on to the Pauling scale. The first scale of electronegativity was developed by Linus Pauling and on his scale zinc has a value of 1.65 on a scale running from from about 0.7 (an estimate for francium) to 2.20 (for hydrogen) to 3.98 (fluorine). The higher the associated electronegativity number, the more an element or compound attracts electrons towards itself. The electronegativity depends upon a number of factors and in particuler as the other atoms in the molecule. Moreover, the electronegativity of an atom is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. This table is a list of electronegativity values of the elements. It can also be used to predict if the resulting molecule will be polar or nonpolar. Electronegativity is used to predict whether a bond between atoms will be ionic or covalent. The most used definition of electronegativity is that an element's electronegativity is the power of an atom when in a molecule to attract electron density to itself. Values for electronegativity run from 0 to 4.
